 The left to right figures shown here are meant to help you see how we can relate the water molecule on the right back to the above drawing of methane. In the drawing below we start off by showing how water has the same number of domains and geometry as methane above, however, we usually see water drawn as the figure on the right. The location of the lone pair around the central atom implies its location in 3D space. As we saw with NH 3 we do not draw a bond to the lone pair but instead, collapse the lone pair directly onto the atom. In H 2O, like NH 3 we have both bonding domains and non-bonding domains. When we have four domains around a central atom our geometry is tetrahedral and the farthest that the four domains can be from each other is 109.5°. The BeF 2 molecule adopts a linear structure in which the two bonds are as far apart as possible, on opposite sides of the Be atom. 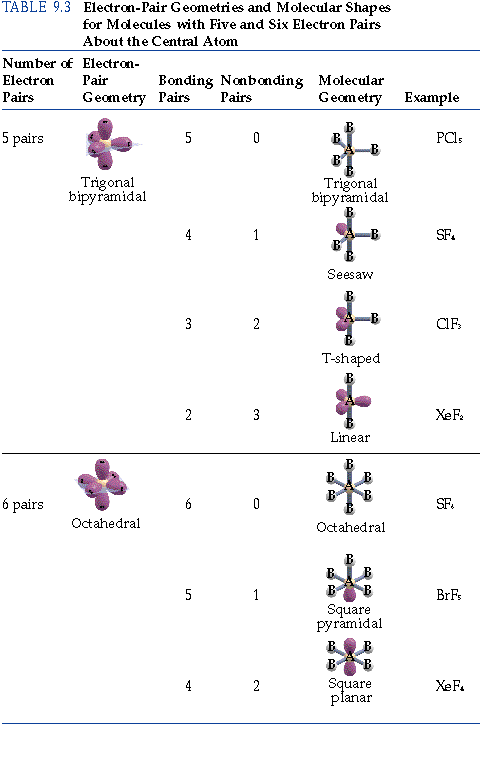
With two bonds and no lone pairs of electrons on the central atom, the bonds are as far apart as possible, and the electrostatic repulsion between these regions of high electron density is reduced to a minimum when they are on opposite sides of the central atom. The Lewis structure of BeF 2 ( ) shows only two electron pairs around the central beryllium atom. The basic electron-pair geometries predicted by VSEPR theory maximize the space around any region of electron density (bonds or lone pairs).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
